Background
Plastics are the bane of the civilised world. With the rise in consumerism, plastics, the invention thought to be the panacea to the problems of the industrialised world, have resulted in unthinkable and unprecedented environmental consequences. The corollary of the overuse of plastic has been far-reaching, and so severe that it threatens the very existence of our planet.
With this, we have embarked on the process of finding biological alternatives to plastic. Our initial biological material we experimented with is chitosan. Given our supervisor, the renowned Professor Javier Fernandez, is a subject matter expert in this field, we decided to tap on his extensive resources and knowledge. We hope to find a suitable alternative which can replicate the properties of plastic, with a lower environmental cost. Our first applications of this, are in consumer disposables, more specifically cups and straws.
The Chemistry and Structure of Chitin
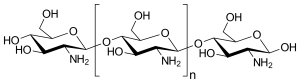
We have attempted to make straws using chitosan, a sugar that is obtained from the hard outer skeleton of shellfish, including crab, lobster, and shrimp.It is made by treating the chitin shells of shrimp and other crustaceans with an alkaline substance, like sodium hydroxide. Chitosan has a number of commercial and possible biomedical uses.
Production of Chitosan Objects
We commence the making of chitosan through mixing dilute acetic acid and chitosan powder. Subsequently, we poured this chitosan solution into the straw moulds we created, or over the cups we have. This is then followed by slotting these chitosan moulds into the dryer at 60 degrees, and then adding NaOH to remove the external H20 groups. This would thereby make the substances hydrophobic.
Results
Our first prototypes, shown below, while rudimentary, are a proof of concept, which we’ll refine further along the production process.


These prototypes, upon contact with water, do not dissolve nor disintegrate. However, they become a lot more soft and malleable, since they lose their mechanical integrity. We intend to heat these straws at 300 degrees to irreversibly remove the inner H20 groups, and subsequently transform this into anhydrous chitosan.
Conclusion
We are in the process of refining and streamlining our production process, to enhance reproducibility and tensegrity of the consumer objects made. We hope to eventually expand the scope of these objects as we make progress, and potentially make them at a lower cost.
By Jaryl Lim
With Sharmayne, Jonathan, and J-Cyn
































